If you’ve been led to believe Lithium-Ion battery packs will cost you more, read on!
Let’s compare some of their key characteristics to help us decide the way forward for your existing or new projects.
Weight
Typically, a standard Lead-Acid battery is three times heavier than an average Lithium-Ion battery of the same capacity. For example, a typical Lead-Acid battery is expected to be 30Kg per KWh, compared to 9Kg per KWh capacity, for a Lithium-Ion Battery. However, in some cases, such as for some electric forklift trucks, the weight of the battery is specified and acts as the vehicle’s counterbalance.
Maintenance

Lead-Acid batteries require relatively high maintenance, since they contain a liquid electrolyte, which is a mixture of sulphuric acid and water. The lead plates in a Lead-Acid battery contain an active material that requires being continually bathed in electrolytes. To find answers to “how often and when” questions, one must refer to the manufacturers’ instructions. Cleaning the terminals and equalising are also important parts of Lead-Acid battery maintenance. Equalising Lead-Acid batteries is a process designed to de-sulphate the battery plates by carrying out a controlled overcharge. Battery plates tend to acquire a sulphate coating over time, which hinders the electro-chemical action between the electrolyte and the plates.
Usable Capacity
Looking at manufacturers’ specification for their maximum allowed daily depth of discharge (dod), it can be seen that Lead-based batteries can be discharged to as much as 50% of their given capacity (e.g. Lead-Acid AGM 15%-30%, Lead-Acid Gel 20%-40% and Lead-Carbon 20% to 50%. In comparison, Lithium batteries can generally be discharged up to 70-80% on a daily basis, and up to 95-100% in backup situations.
Charging and Discharging
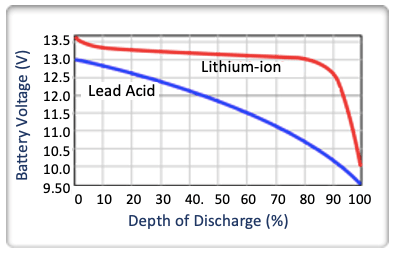
Lithium-Ion chemistries can accept a faster rate of charge current, compared to Lead-Acid batteries. Typically,Lithium-Ion batteries may charge as quickly as in a few minutes, while equivalent Lead-Acid batteries could take over 10 hours, depending on the capacity of the battery.
Lithium provides a constant power for loads, throughout the discharge cycle . Lead-Acid starts strong, and then dissipates, as shown in the graph.
Temperature
It’s a recognised industry standard that Lithium-Ion batteries, while their storage temperature ranges from –40°C to 50°C (–40°F to 122°F), and their operating temperature from +10°C to +55°C (50°F to 131°F), charging should take place only at a battery temperature of +5°C to +45°C (+41°F to +113°F). Teviot Technology Inc. is working on a series of ultra-smart LiFeSo4 ( LFP ) battery pack technologies to extend these limitations. For all your lithium battery customisation requirements, please talk to us.
For Lead-Acid batteries, the allowable storage temperature ranges from –40°C to 50°C (–40°F to 122°F), while their charging and operating temperature ranges are from –20°C to 50°C (68°F to 122°F).
Installation
To prevent extremely corrosive acid mixture leakage, Lead-Acid batteries, unless of the sealed type, can only be installed in an upright -orientation. However, as Lithium-Ion battery cells are always sealed, orientation is not an issue.
Safety
There are a number of chemistries used in Li-Ion batteries. Teviot Technology Inc uses Lithium Iron Phosphate, LiFeSO4 (LFP) cells, known to be the safest and most stable type available. LFP does not exhibit the energetic thermal runaway that metal oxide Lithium-Ion cells experience, which has occasionally led to battery fires.
While Lead-Acid batteries present low fire risks, they are capable of storing large amounts of energy and often utilise a volatile or combustible electrolyte and thus are still somewhat prone to fires and explosions. In addition, at any time dealing with Lead-Acid maintenance, full protection gear has to be used by the technician.
Life Span
Teviot’s choice of Lithium-Ion battery cell chemistry, LiFeSO4, has become known as one of the most robust and long-lasting battery chemistries available. These batteries last ten years or more, if correctly maintained, with cycle durability ranging from 2,000 to12000 cycles.
A deep-cycled Lead-Acid battery delivers 100–200 cycles before a gradual decline begins. Replacement should occur when the capacity drops to 70 or 80 percent.
Space & Energy Density
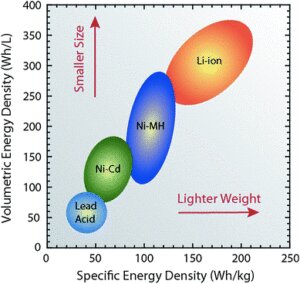
With their fast-charging time, Lithium Batteries reduce or eliminate the need for spare batteries. They also don’t require dedicated charging rooms. Additionally, comparing energy densities, Lead-Acid batteries have an 80-90 Wh/L energy density, compared to 250-670 Wh/L for Lithium-Ion batteries.
A diagram of the specific energy density and volumetric energy density of various battery types. Li-ion batteries are ahead of most other battery types in these respects. (Roberta A. DiLeo, Rochester Institute of Technology)
Costs
The above-mentioned general maintenance is one of the greatest operational costs of Lead-Acid batteries, as it includes topping up with water, maintaining the filling system and removing oxide deposits from the elements and terminals. However, some of the hidden costs of Lead-Acid batteries should also be considered and these include infrastructure costs, gas disposal and water demineralisation. Lead-Acid batteries release gasses while being charged, and therefore have to be charged in dedicated areas. In addition, the gasses released must be removed by a special ventilation system. Demineralisation is a necessary treatment for water used to top-up Lead-Acid batteries. These costs become significant for medium to large fleets. In addition, spare Lead-Acid batteries are another operational cost.
While the initial costs of purchasing Lithium-Ion batteries are higher than for Lead-Acid batteries; However, considering all the secondary costs and complexities that must be considered, Lithium-Ion batteries have an estimated project cost of $469 per kWh, compared to $549 per kWh for Lead-Acid, according to the U.S. Department of Energy’s 2019 Energy Storage Technology and Cost Characterization Report.
Environmental Effects
Lead-Acid batteries pose a potential threat to human health and the environment, if improperly discarded. The two main components of these batteries are sulphuric acid and lead. Both lead and sulphuric acid can contaminate solid ground and ground water. Sulphuric acid is highly corrosive, and lead has been linked to adverse health effects in humans, particularly children. UNC Environmental Facts Sheet.
As mentioned previously, Teviot Technology Inc. uses Lithium Iron Phosphate (LiFePO4 or LFP battery) for their battery packs. These type of battery cells, use lithium ferric phosphate as the cathode material, and a graphitic carbon electrode with a metallic backing as the anode. LFP does not contain nickel or cobalt. Lithium-ion battery chemistries which use Cobalt or Nickel for the cathode have caused concerns for their negative impact on the environment and human health.
Conclusion
In summary, looking at weight, maintenance, usable capacity, temperature, installation, safety, charging, discharging, life span, energy density, cost and environmental impact, the choice becomes clearer; and in favour of Lithium Iron Phosphate, LFP batteries. Lithium-based batteries are quickly becoming a preferred replacement for the 150-year-old technology of Lead-Acid batteries. For all your eMobility Lithium-Ion battery requirements, please contact Teviot Technology Inc.
Technology executive with an extensive track record in electronics, firmware and software development, production, business development, strategic planning and marketing, in the automotive, medical, aerospace, educational, IoT, and telecom fields. I have formed and led new companies and engineering departments, managing technology development programs and projects from initial concept right through to production. I have increased operational efficiencies through initiating and establishing new processes, tools and training. I am a creative thinker who relishes finding new approaches to solving problems. A positive, motivating leadership style, establishing effective working relationships with my peers, while building strong rapport with my external and internal customers, have all been key contributing factors toward my career successes.

